Hey PCR enthusiasts,
Here are some updates from Bento Lab!
Methods and Techniques
DNA Extraction from Microorganisms via Hot Alkaline Fracturing of Cell Walls
Microorganisms with tough cell walls can offer challenges for DNA extraction. However, Liu et al. (2022) propose a simple, affordable, and rapid method that leaves DNA intact but available for PCR within damaged and fractured cells. The cells can then be pelleted, resuspended, and used directly for the amplification of DNA fragments of up to 6,500 bp and possibly more!
The authors call the method “PBC” (potassium hydroxide + boiling + centrifugation). It involves:
- Boiling for 10 mins in 0.1 M KOH
- Centrifugation
- Discarding the supernatant
- Resuspending the sediment in 100 μL of sterile water
- Using the cell suspension directly for PCR
The PBC method is similar to other hot alkaline lysis methods, but it uses KOH rather than other alkalis, has a higher lysis temperature (100 °C vs 94 °C or lower), and uses suspended cell sediment as a DNA source rather than free DNA released into the extraction buffer.
The authors think that PBC extractions could be easily scaled up for routine processing of large numbers of samples, and had already tested it on hundreds of PCRs from smut fungus clones at the time of publication. However, it does rely on the use of a centrifuge which can be a limiting factor in throughput.
The study is a very detailed investigation with lots of extra detail, so well worth a read! You can find the article here:
Product Highlight
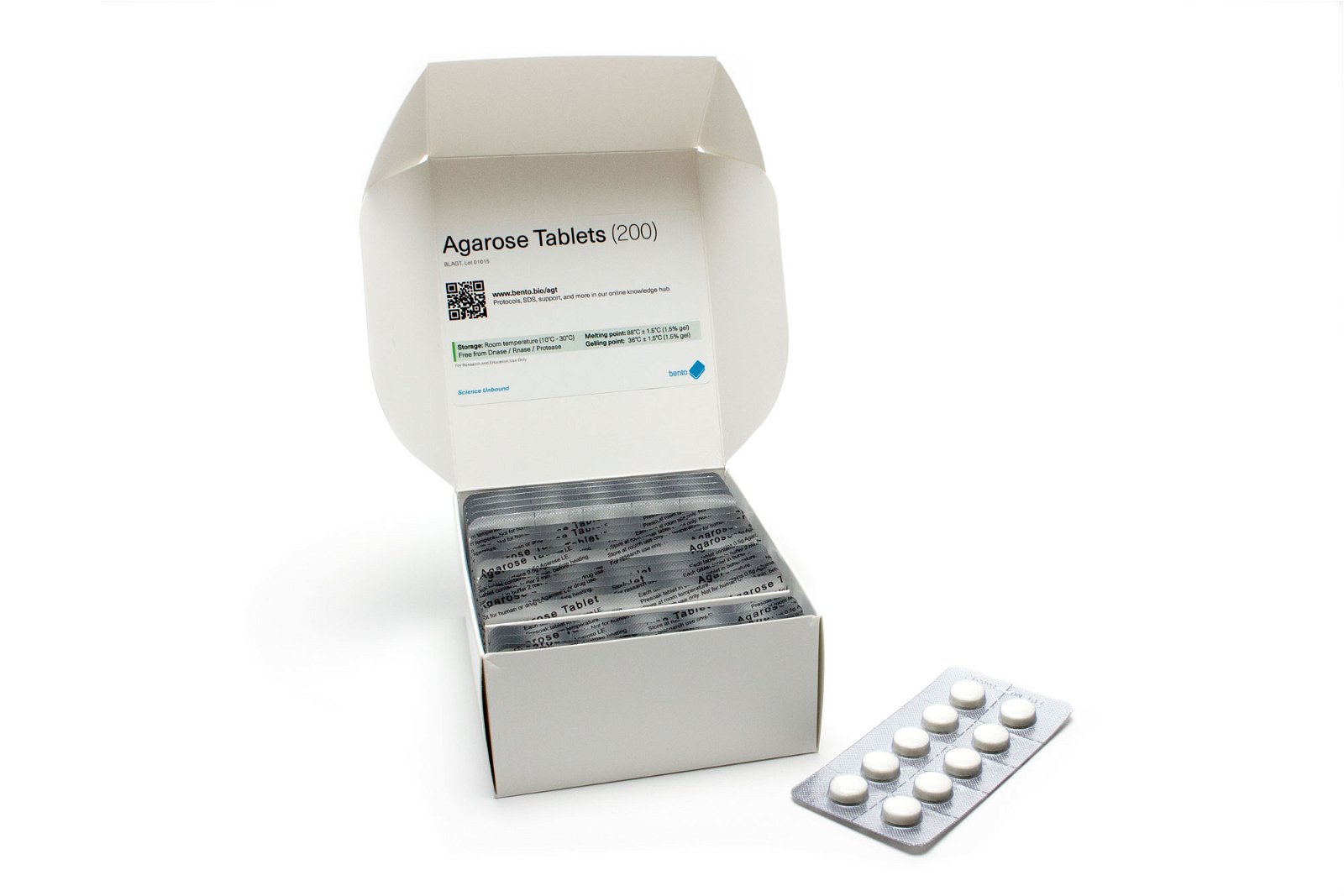
Agarose Tablets
Agarose tablets are a great way of making agarose gels for gel electrophoresis. No weighing, no spills, no waste, and consistent gel concentrations. They make gel preparation fast and hassle-free!
For a 1% gel, just drop a 0.5g tablet into 50 mL of TBE or other electrophoresis buffer. The tablets take a few minutes to disintegrate in the buffer, and you can then melt them using a microwave or other heat source (e.g. a rice cooker or camping stove) until they are fully dissolved and molten.
This ease of use is particularly useful in educational settings, fieldwork, or for those new to electrophoresis.
If you want to try agarose tablets, we stock them in small quantities of 10 for $5.99 ($0.59 a gel), or you can buy a box of 200 for $72.99 ($0.37 a gel). You can find them in our store here.
If you have any other great products you’d like us to stock, please drop us a message!
Articles We Love
Mass Forensic Genotyping of Illegally Traded Pangolins
PCR and sequencing-based methods are essential tools in the identification and tracing of illegally traded animal or plant products, but large seizures can prove challenging and costly to process for provenance-tracing and prosecution.
A new approach, by Yeo et al. (2023), used Oxford Nanopore sequencing to significantly increase capacity while reducing costs, in the (probably) first example of high-throughput rapid genotyping and provenance tracing for wildlife forensic genotyping. The authors were able to DNA barcode an impressive 2346 scales from a large seizure of pangolin skins, and process them at a cost of less than 1 USD per sequence!
The authors used several optimisations to keep the cost down and to increase throughput, including:
- Use of the HotSHOT DNA extraction method for quick high-throughput extractions
- Using dithiothreitol (DTT) with HotSHOT to improve DNA yield and amplification success
- Tagged primers for sample multiplexing
- High-throughput sample processing using 96 well plate format workflows
Once the scales were sequenced and analysed, Yeo et al. (2023) were able to identify the scales to species and distinct haplotype level, matching the haplotypes to known phylogeographical populations.
The data indicated poaching primarily from Western-central Africa, Western Africa, and Gabon. The number and diversity of haplotypes poached indicated that skins were being centrally pooled before being shipped overseas. The scale of collections also indicated that continued intensive poaching may also impact the viability of local populations and create population bottlenecks.
Importantly, the methods developed and demonstrated by the authors represent a rapid, scalable, and cost-efficient genotyping framework that can be adapted for any large seizure of wildlife products.
You can find the article here:
The methods shown here could also be done on a smaller scale using Bento Lab, using a 32 well rather than 96 well plate format. This could make rapid-response genotyping and provenance tracing for wildlife forensics even more portable and adaptable!
We would love to hear from anyone interested in (or already doing) this kind of work!



