Overview
Many birds are sexually monomorphic, which means that their sex can not be identified based on appearance. Instead, DNA testing can be used based on male and female birds having a different combination of the bird sex chromosomes Z and W.
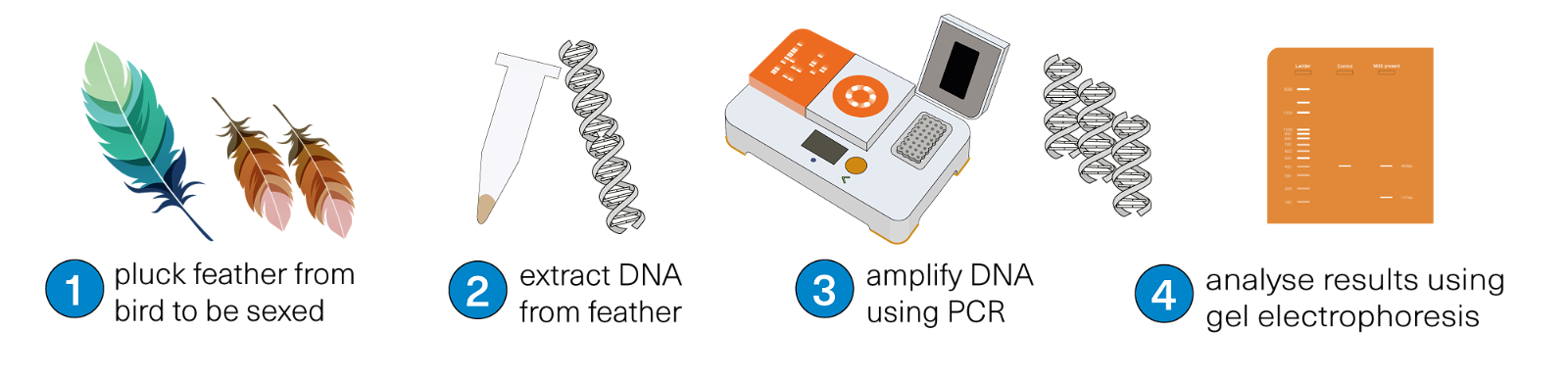
This collection of bird sexing workflows shows how to extract DNA from feathers, and how to identify the sex of birds using PCR and gel electrophoresis. Two bands indicate a female bird, one band indicates a male bird.
Are you interested in DIY bird sexing?
We are passionate about making the power of PCR available to anyone. Identifying the sex of birds can be essential for bird owners, breeders, conservationists, and many others.
If you are interested in carrying out this workflow for yourself, we would love to hear from you! You can find all of the resources and information on these pages, and if you have questions, we are happy to answer them.
What will you need for this workflow?
The reagents, consumables and equipment you need in order to follow the DNA Extraction from Feathers and Bird Sexing Protocol are listed below.
Reagents
- HotSHOT DNA Extraction Kit
- 5x FIREPol® Master Mix Ready To Load or 5x HOT FIREPol® Blend Master Mix Ready to Load
- PCR Grade Water
- Bird Sexing Primer Mixes
- Agarose Tablets
- GelGreen® DNA Stain
- 100 bp DNA Ladder
- 10X TBE buffer (to be diluted to 0.5X)
- Deionised water (not included)
Consumables
- Plucked feather(s)
- Disposable scalpels or razor blades (not included)
- Nitrile gloves (not included)
- Paper towel (not included)
- 2-200μl Pipette Tips (1 rack)
- PCR tubes
Equipment
- Bento Lab
- Glass beaker or conical flask (not included)
- 2-20μl Adjustable Pipette
- 20-200μl Adjustable Pipette
- Microwave
Scientific Background – How does it work?
Birds have two sex chromosomes known as Z and W. These are similar to the X and Y sex chromosomes in humans – each inherited from one parent. Male birds have two copies of the Z chromosome (ZZ) and females have one copy of the Z chromosome and one copy of the W chromosome (ZW). This is the opposite of the situation to humans, where most biological females have two copies of the X chromosome (XX) and most biological males have an X and a Y chromosome (XY).
This PCR workflow involves the CHD1 (Chromodomain Helicase DNA Binding Protein 1) genes, which are present on the W and Z sex chromosomes of birds as two variants – CHD1-W (on the female-specific W chromosome) and CHD1-Z (present in males and females on the Z chromosome). These genes are homologous (almost identical in structure and function) but contain introns (DNA that is removed by RNA splicing before it is translated into mature RNA) that usually differ in length between sexes within a species and between species.
This difference in length allows both sexes to be determined using a simple assay based on intron length differences. Once DNA has been extracted, regions within the CHD1-Z and CHD1-W (if present) genes can be amplified using the polymerase chain reaction (PCR), and the amplified DNA visualised on an electrophoresis gel. If only one band is present, this suggests that only one CHD1 coding variant is present (CHD1-Z), which would be expected for a male with a ZZ chromosome pair. If two bands are present, this suggests that both CHD1-Z and CHD1-W genes are present, which would be expected for a female with a ZW chromosome pair.
The advantage of amplifying both variants (if present) together is that the band produced by the CHD1-Z should always be visible – it will only be absent if the DNA extraction or PCR fails. It therefore acts as an individual positive control for each specimen.
What are the possible results of this PCR test?
There are two possible results for most birds:
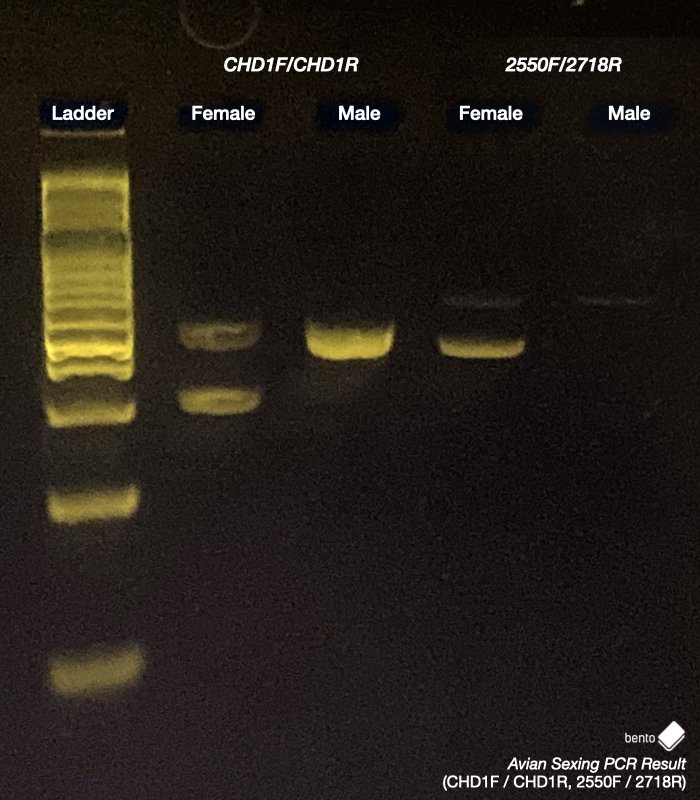
Female: One copy of the CHD1-Z gene is present on the Z chromosome, and one copy of the CHD1-W gene is present on the W chromosome. The target amplified regions of these genes are generally different in length and will be visible on an electrophoresis gel as two distinct bands.
Male: Two copies of the CHD1-Z gene are present – one on each of the Z chromosomes. The amplified regions of these genes are almost always identical in length and will be visible on an electrophoresis gel as a single band.
Case study: Domestic chicken (Gallus gallus domesticus)
There are two primer sets that are commonly used for bird sexing as they can successfully sex birds belonging to many, but not all, taxonomic groupings. These are CHD1F paired with CHD1R, and 2550F paired with 2718R.
When these primers are used to amplify regions of the CHD1-W and CHD1-Z genes from DNA extracted from a female and a male chicken, they produce different amplicons for each sex as can be seen below: two distinct bands for the female specimen, and only one band for the male specimen.
Which primer set do I need?
You can decide which primer set is most suitable based on what species you want to sex, or try multiple primer sets to see which work best with a species. Each primer set works for a range of species listed below. This list is not exhaustive and should be taken as a guide only.
For any species, we suggest that users try one or more of the following steps to help select the most appropriate primers for sexing:
- Consult the relevant literature to determine what primers have been shown to work well with particular species in the past. In some cases additional primer sets beyond the three covered here may be needed. For example, anyone interested in sexing parrots should read our blog post on a new strategy for sexing different parrot species here, and Table 1 of Kroczak et al., 2021 for information on which primers produce reliable results for which species.
- Validate the assay with known male and female birds of that species to ensure a reliable result.
- Test all three primer mixes on a species to achieve a consensus result that can then be used to identify the most appropriate primer sex for that species. In rare cases none of the primer sets we supply may be appropriate, and additional species specific primer sets may be required.
- Test primers on a large enough sample size of a species to be sure to detect both male and female birds within that set. If the results indicate the presence of both male and female samples then the primers used are probably appropriate.
| Species | Scientific Order | Primer Set 1 (CHD1F/CHD1R) | Primer Set 2 (2550F/2718R) | Primer Set 3 (P0/P2/P8) |
|---|---|---|---|---|
| Landfowl, domestic chicken | Galliformes | ✓ | ✓ | |
| Pelican | Pelecaniformes | ✓ | ✓ | |
| Flamingo | Phoenicopteriformes | ✓ | ✓ | |
| Birds of prey, hawks, eagles, vultures | Accipitriformes | ✓ | ✓ | |
| Waterfowl | Anseriformes | ✓ | ✓ | |
| Songbirds | Passeriformes | ✓ | ||
| Stork, kestrel | Ciconiiformes | ✓ | ||
| Pigeons | Columbiformes | ✓ | ||
| Parrots | Psittaciformes | ✓ (also see here) | ||
| Kookaburra | Coraciiformes | ✓ | ||
| Cranes, adzebills | Gruiformes | ✓ |
Costing Your Bird Sexing Workflow
We’ve put together a spreadsheet calculator to help users of these protocols better understand the costs and resources needed to sex a given number of birds. If you have any questions or feedback about the spreadsheet, please get in touch. You can download the spreadsheet calculator here.

